Toxicology Laboratory Services Manual
Table of Contents
Specimen Submission Recommendations
Postmortem Specimen Submissions
Antemortem Specimen Submissions
Appendix A: Quick Reference Guide for Submitting Antemortem Specimens
Mission
The discipline of Forensic Toxicology at the Sedgwick County Regional Forensic Science Center (RFSC) involves the analysis of biological samples to determine the presence and/or absence of drugs and other substances (e.g., ethanol, methamphetamine, prescription drugs, etc.) in postmortem and antemortem cases.
Postmortem analyses are used to assist the Sedgwick County District Medical Examiner’s Office in determining the cause and manner of death. Support for coroners outside of Sedgwick County is provided according to the current fee schedule.
Antemortem analyses are used to provide information to law enforcement in cases involving driving under the influence (DUI), driving under the influence of drugs (DUID), and/or drug facilitated sexual assaults (DFSA).
Quality Statement
The quality system applies to all work performed in the laboratory’s facility and has been designed to meet the requirements for laboratory accreditation and, where appropriate, closely follow standards established by the American Board of Forensic Toxicology, the Society of Forensic Toxicologists and the American Academy of Forensic Sciences.
Specimen Submission Recommendations
Specimen submission recommendations are summarized in:
Appendix A: Quick Reference Guide for Submitting Antemortem Specimens
Postmortem Specimen Submissions
Biological specimens collected during autopsies conducted at the Regional Forensic Science Center may be submitted to the toxicology laboratory for analysis.
1. Specimen Types
a. Specimens routinely accepted may include blood [hospital, heart, femoral, subclavian, cavity/chest], serum [hospital], chest fluid, vitreous, urine, gastric contents, brain, liver, kidney, spleen and muscle. Additional specimens may be submitted if necessary.
b. Physical evidence collected at the death scene or during autopsy may be submitted for analysis by the RFSC Drug Identification Unit.
2. Specimen Documentation
a. The lab order must be completed and included with the specimen(s).
- The case pathologist is responsible for indicating the investigative and medical history and toxicology indications, drug treatment(s) and/or chemical exposure(s), postmortem findings and the toxicology analysis desired.
3. Specimen Containers
a. All “wet” specimens [e.g. blood, urine, tissue, etc.] collected from autopsy are to be submitted in a properly sealed clear plastic biohazard bag. Each specimen in the bag must be submitted in a specific manner, which generally is a closable glass or plastic tube or specimen container that may contain an additive. Specimens may be received in additional manners.
b. All “dry” physical evidence [e.g. drug paraphernalia] collected from a death scene or during an autopsy are to be submitted in a properly sealed plastic zip type, paper bag or envelope. Physical evidence may be received in additional manners.
c. All syringes submitted to the laboratory for analysis must be placed inside a puncture proof container prior to placing inside of the properly sealed outer container described above.
4. Specimen Volume
a. The ideal minimum volume for performing all testing is:
| Sample Type | Minimum Volume |
|---|---|
| Blood | 10 mL |
| Urine | 10 mL |
| Vitreous | 2 mL |
| Chest Fluid | 10mL |
| Tissue | 10g |
Note: These volumes are ideal. The laboratory understands that sometimes less volume is all that is available. The laboratory will work to determine testing priorities in cases with specimens that have limited sample volume.
5. Specimen Labeling
a. Using non-gel ink or a label, the specimen containers should be labeled, at a minimum, whit the following information:
- Decedent's Name (if unknown, indicate as such);
- Pathology Case Number;
- Date of Autopsy;
- Pathologist; and,
- Specimen Type.
6. Evidence Labeling
a. Properly sealed packages should bear, at a minimum, the date the seal was applied and the initials of the person that sealed the evidence.
b. The date and initials should be placed across the seal, which includes tape or heat seal.
Antemortem Specimen Submissions
Driving Kits (DUI/DUID)
Biological specimens collected during the investigation of a driver suspected to be under the influence of drugs and/or alcohol may be submitted for analysis.
1. Specimen Documentation
a. Antemortem specimens submitted for analysis must be pre-logged. This can be completed prior to evidence drop off via the RFSC Laboratory Information Management System (LIMS) website (www.rfsc.sedgwickcounty.org/fawebportalprod) or at the RFSC Evidence Submission window kiosk.
- The Law Enforcement Agency is responsible for indicating the investigative and medical history and any known toxicology indications, drug treatment(s) and/or chemical exposure(s), investigative findings, and the toxicology analysis desired.
- The submitting law enforcement agent is responsible for indicating each specimen submitted for analysis. They are also responsible for properly initiating the chain of custody.
2. Specimen Containers
a. Blood: The recommended container for blood collection is a 10 mL gray top evacuated blood collection tube containing both a preservative and an anticoagulant. The preservative helps prevent deterioration of drug(s) that may be present in the specimen and the anticoagulant prevents the specimen from clotting.
- Blood collected in a different type of tube may be tested on a case-by-case basis.
b. Urine: The recommended container for urine collection is a plastic urine specimen container that is tolerant to transportation. Preservatives are not required.
Note: Both blood collection tubes and urine specimen containers must be submitted in a container (e.g., zip-top bag, blood draw kit, envelope, or box). The outermost container may be paper as long as the proximal container to the specimen container(s) is plastic.
3. Specimen Volume
a. The ideal minimum volume for performing all testing is:
| Case Type | Minimum Volume |
|---|---|
| Blood DUI | 2 mL |
| Blood DUID | 30 mL (3 xx10 mL gray top tubes) |
| Urine DUID | 20 mL |
Note: These volumes are ideal. The laboratory understands that sometimes less volume is all that is available. The laboratory will work to determine testing priorities in cases with limited sample volume.
4. Specimen Type
a. Always collect blood for cases involving driving performance and/or impairment.
b. Collect urine, in addition to blood, for cases when heroin usage is suspected.
c. If the blood alcohol content is ≥ 0.100 gm%, additional testing for drugs will not be performed. Exceptions may be allowed after discussion with the Lab Director or Toxicology Laboratory Manager.
d. Urine is not routinely tested for volatiles in suspected DUI/DUID cases.
5. Specimen Labeling
a. Using non-gel ink and the labels provided in the DUI/DUID Blood Draw Kit, the specimen containers should be labeled, at a minimum, with the following information:
-
- Subject's Name;
- Law Enforcement Agency Case Number;
- Date of Collection;
- Time of Collection; and,
- Address.
b. Other suitable labels may be used on the specimen containers as long as the above information criteria are met.
6. Specimen Seals
a. A tamper resistant tape seal should be placed on the outermost container.
- Alternatively, containers may be submitted in a heat sealed plastic bag.
b. The person sealing the outermost container should initial and date the seal.
7. Specimen Storage
a. In order to maintain integrity and preservation, specimens should be kept secure and refrigerated until submitted to the laboratory.
8. Chain-of-Custody
a. Chain of custody is initiated via the RFSC LIMS upon evidence submission at the Evidence Window.
Note: Additional information may be useful and requested to complete analysis.
Drug Facilitated Sexual Assaults (DFSA)
Biological specimens collected during the investigation of a suspected drug facilitated sexual assault (DFSA) may be submitted for analysis.
1. Specimen Documentation
a. Antemortem specimens submitted for analysis must be pre-logged. This can be completed prior to evidence drop off via the RFSC Laboratory Information Management System (LIMS) website (www.rfsc.sedgwickcounty.org/fawebportalprod) or at the RFSC Evidence Submission window kiosk.
- The Law Enforcement Agency is responsible for indicating the investigative and medical history and any known toxicology indications, drug treatment(s) and/or chemical exposure(s), investigative findings, and the toxicology analysis desired.
- The submitting law enforcement agent is responsible for indicating each specimen submitted for analysis. They are also responsible for properly initiating the chain of custody.
2. Specimen Containers
a. Blood: The recommended container for blood collection is a 10 mL gray top evacuated blood collection tube containing both a preservative and an anticoagulant. The preservative helps prevent deterioration of drug in the specimen and the anticoagulant prevents the specimen from clotting.
- Blood collected in a different type of tube may be tested on a case-by-case basis.
b. Urine: The recommended container for urine collection is a plastic urine specimen container that is tolerant to transportation. Preservatives are not required.
Note: Typically, DFSA specimens are submitted in a Drug Facilitated Sexual Assault Blood and Urine Specimen Collection Kit. However, law enforcement agencies may prepare their own kits as a substitute, as long as they closely mimic the standard collection kit.
| Sample Type | Minimum Volume |
|---|---|
| Blood | 20 mL (2 x 10 mL gray top tubes) |
| Urine | 20 mL |
Note: These volumes are ideal. The laboratory understands that sometimes less volume is all that is available. The laboratory will work to determine testing priorities in cases with limited sample volume.
4. Specimen Type
a. Always collect blood and urine for DFSA cases.
5. Specimen Labeling
a. Using non-gel ink or labels, the specimen containers should be labeled, at a minimum, with the following information:
- Subject’s Name;
- Law Enforcement Agency Case Number;
- Date of Collection; and,
- Time of Collection.
b. Additional labeling of specimen containers may be provided as required by standardized collection kits, as long as the above information criteria are met.
6. Specimen Storage
a. A tamper-resistant tape seal must be placed on the outermost container. Specimen containers may be further sealed with tamper-resistant tape.
- Alternatively, containers may be submitted in a heat sealed plastic bag.
b. The person sealing the outermost container should initial and date the seal.
7. Specimen Storage
a. In order to maintain integrity and preservation, specimens should be kept secure and refrigerated until submitted to the laboratory.
8. Chain-of-Custody
a. Chain of custody is initiated via the RFSC LIMS upon evidence submission at the Evidence Window.
Note: Additional information may be useful and requested to complete analysis.
Analytical Results
- The RFSC Toxicology Laboratory issues final postmortem toxicology reports to the Sedgwick County Chief Medical Examiner or his/her deputies.
- The RFSC Toxicology Laboratory issues final antemortem toxicology reports to the submitting law enforcement agency.
- Release of additional copies of toxicology reports will be in accordance of RFSC policies and any applicable law.
- A “not detected” result on a toxicology report does not necessarily mean drugs were not present or ingested. It could mean that they are present below the laboratory’s reporting criteria.
Specimen Return
- Postmortem specimens are retained for approximately three years, after which they are disposed. Specimens may be retained for longer than three years if required by the courts.
- Antemortem specimens are returned to the submitting law enforcement agency after the laboratory report has been issued.
Appendix A
Appendix A: Quick Reference Guide for Submitting Antemortem Specimens
Recommended DUI/DUID Kit Packaging
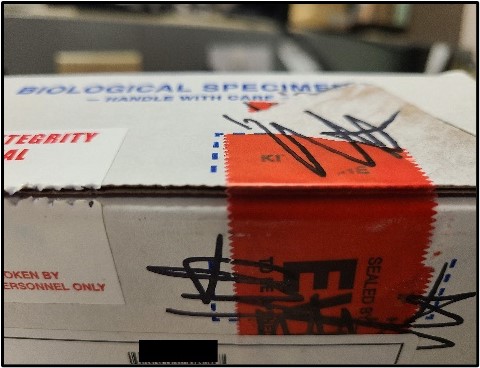
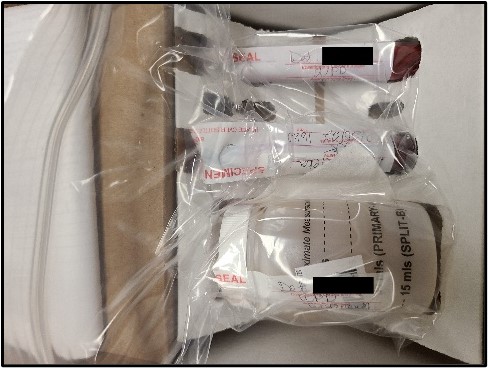
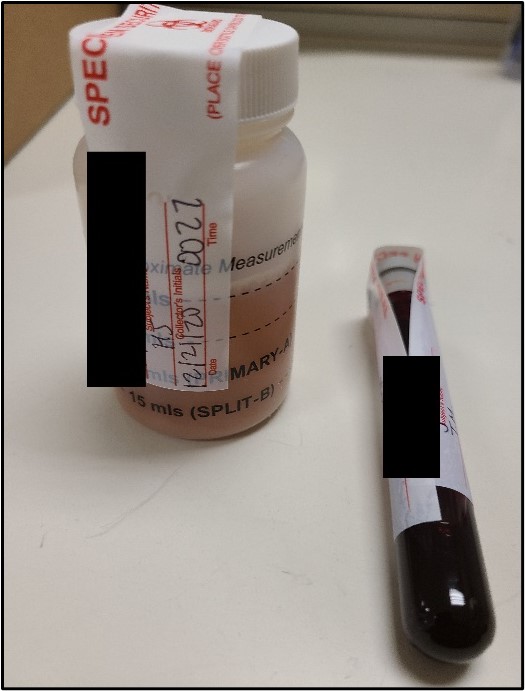
Below are instructions for the ideal way to package DUI/DUID Blood Draw Kits for submission to the Toxicology Laboratory for analysis. Adhering to these instructions will ensure evidence integrity and expedite case analyses.
It is recognized that not all DUI/DUID Blood Draw Kits will be submitted following these instructions exactly. In those instances, the case working scientist will evaluate the packaging to ensure that evidence integrity has been maintained. The laboratory may contact the submitting law enforcement agent if need be to facilitate analysis.
Photos of recommended condition(s) to submit blood kits to facilitate analysis.





Recommended DFSA Kit Submission
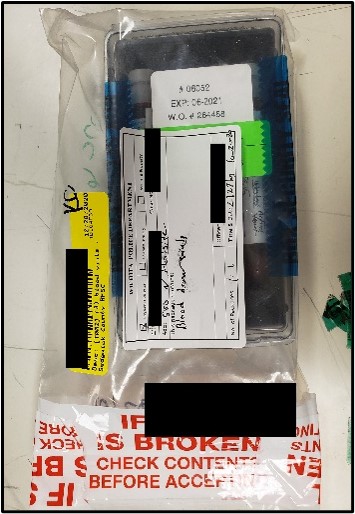
Below are instructions for the ideal way to package DFSA Kits for submission to the Toxicology Laboratory for analysis. Adhering to these instruction will ensure evidence integrity and expedite case analyses.
It is recognized that not all DFSA Kits will be submitted following these instructions exactly. In those instances, the case working scientist will evaluate the packaging to ensure that evidence integrity has been maintained. The laboratory may contact the submitting law enforcement agent if need be to facilitate analysis.
Photos of recommended condition(s) to submit DFSA Kits to facilitate analysis.